 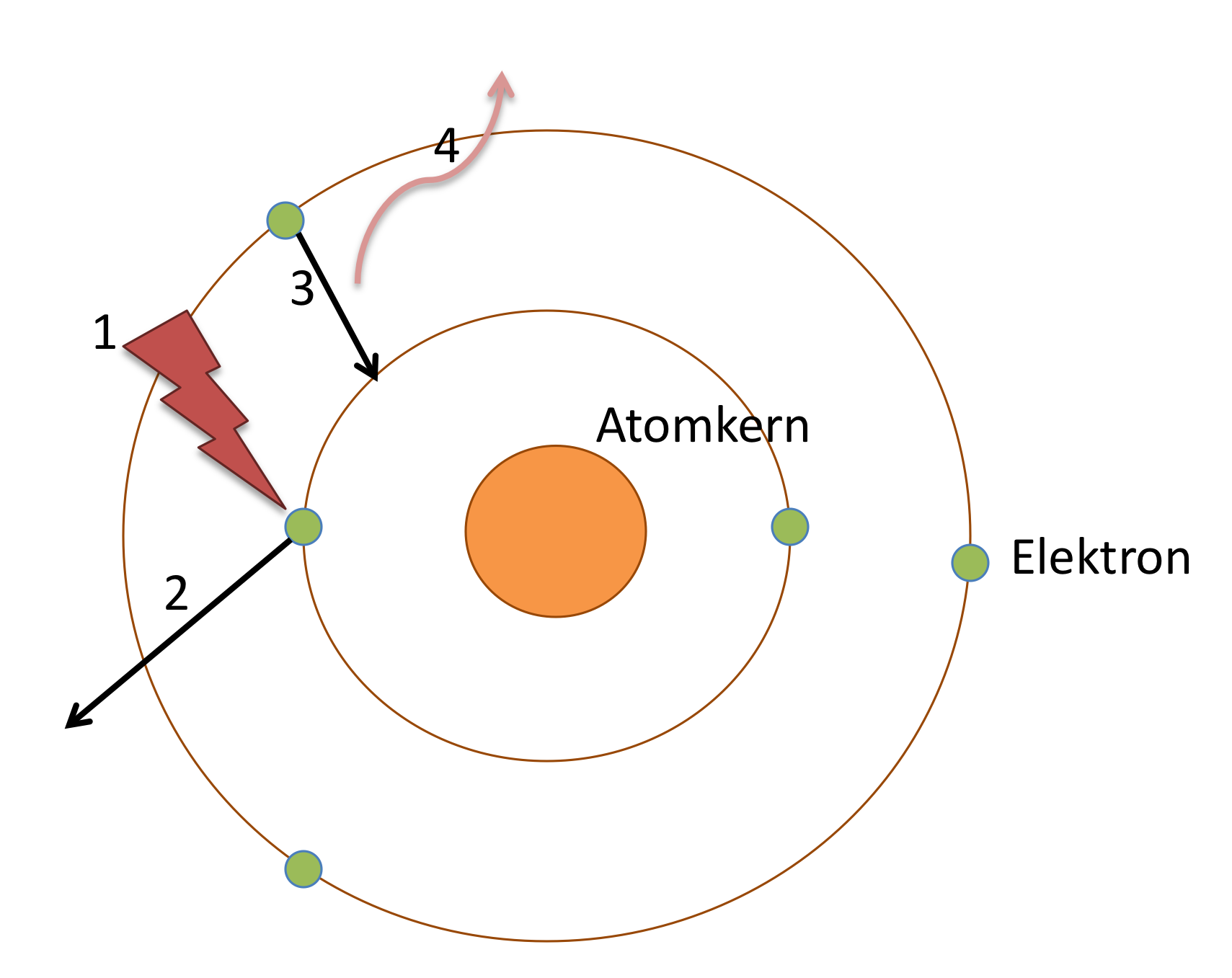
This concept provided a framework for understanding electron behavior within atoms. According to the model, electrons occupy quantized energy levels and remain in stable orbits unless they absorb or emit energy during transitions between energy levels. Understanding Electron Stabilityīohr’s model introduced the concept of stable electron orbits. By considering the electrostatic attraction between the positively charged nucleus and the negatively charged electron, the model accurately predicted the energies of different electron orbits. In this article, we will explore the advantages and disadvantages of Bohr’s atomic model, delve into calculations of radius, energy, velocity, and time period of electrons in atoms, and address frequently asked questions related to this influential model.īohr’s model allowed for the calculation of energy levels for electrons in hydrogen and hydrogen-like atoms. This model provided insights into the behavior of electrons in atoms, explaining their stable orbits and emission/absorption of energy. Can Bohr's model explain the emission and absorption spectra of elements other than hydrogen?īohr’s atomic model, proposed by Niels Bohr in 1913, revolutionized our understanding of atomic structure.How does Bohr's model explain the stability of atoms?.How does Bohr's model relate to the modern quantum mechanical model of the atom?.What is the significance of the principal quantum number in Bohr's model?.Does Bohr's atomic model still hold significance today?.Why did Bohr's model fail to explain the behavior of complex atoms?.Lack of Explanation for Chemical Bonding. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
